Volume 1, Issue 2
Article Type: Research Article
The relationship between cerebral small vessel disease and presbyvestibulopathy: Our experience
Paolo Gamba1*; Gabriella Licursi1; Luciano Ketty1; Claudio Battistuzzi1; Luca Cavalcanti2; Arianna Daquino3; Umberto Pignatelli1
1Department of Otorhinolaryngology - Head and Neck Surgery LAB of Clinical and Instrumental Vestibology, Poliambulanza Foundation Hospital Brescia, Italy.
2Department of Sense Organs, Sapienza University of Rome, Italy.
3Department of Surgical Sciences, ENT Unit, University of Turin, Italy.
*Corresponding author: Paolo Gamba
Department of Otorhinolaryngology - Head and Neck Surgery,
LAB of Clinical and Instrumental Vestibology, Poliambulanza Foundation Hospital Brescia, Italy.
Email ID: paolo-gamba@libero.it
Received: Jun 13, 2025
Accepted: Jul 07, 2025
Published Online: Jul 14, 2025
Journal: Annals of Gerontology and Geriatrics
Copyright: Gamba P et al. © All rights are reserved
Citation: Gamba P, Licursi G, Ketty L, Battistuzzi C, Cavalcanti L, et al. The relationship between cerebral small vessel disease and presbyvestibulopathy: Our experience. Ann Gerontol Geriatr Res. 2025; 1(2): 1011.
Abstract
Objective: Presbyvestibulopathy is defined as a condition of persistent dizziness or unsteadiness associated with findings on laboratory tests that are established for bilateral vestibulopathy. Magnetic Resonance Imaging (MRI) features of White Matter Lesion (WMLs) are correlated with dilatation of perivascular spaces, especially in the frontal, and parietal subcortical area, and around the ventricular system. The aim of our work was to investigate the correlation between small WMLs and Presbyvestibulopathy (PVP).
Methods: A total of 48 patients were enrolled in the present study. Vascular risk factors were evaluated for each patient. All subjects had an otoneurological examination for the PVP instrumental diagnostic criteria, only values from the vHIT were selected (aVOR gain threshold as <0.95 and >0.6). All participants underwent 1,5 Tesla MRI scans. White Matter Lesions hyperintensities on 48 patients, MRI T2-weithted and Fluid Attenuated Inversion Recovery sequences were rated according to the Fazekas scale.
Results: Results suggest that the complaint of the high prevalence of frontal lobe and parieto-insular cortex of WMLs. The estimates of peak location result from analysis of several neuroimaging of the white matter lesion can be subdivided in five major areas: frontal lobe, deep periventricular, parieto-insular vestibular cortex, corona radiate and thalamus. Our data reveal the functional significance of the prefrontal cortex in modulating anxiety and other emotional behaviors through its connections to the amygdala.
Conclusions: Healthcare practitioners need increased awareness of the presbyvestibulopathy pathophysiology and its possible impact, as small White Matter Lesions in elderly patients can no longer be considered an incidental, insignificant findings.
Keywords: Chronic subjective dizzines; Presbyvestibulopathy; Small white matter lesions.
Introduction
Several terms have been used to describe an age-related vestibular deficit, such as presbyequilibrium, presbyvertigo, presbyastasis, and Presbyvestibulopathy (PVP). The latter has been recently defined by the ad hoc committee of Bàràny Society [1]. Therefore, PVP is defined as a chronic vestibular syndrome clinically characterized by unsteadiness, gait disturbance and/or recurrent fall (Table 1). PVP is consistent with other age-related sensory losses such as presbycusis and presbyopia. Since threequarters of individuals affected by PVP have co-existing anxiety or depressive symptoms an overlap with dizziness of psychogenic origin should be considered [2]. The existence of cognitivevestibular interaction is supported by the presence of neuronal projections between the vestibular centers in the brainstem and both the cerebral and cerebellar cortex [3-5]. Over the past few years, there has been growing interest in the literature regarding areas in cerebral white matter that appear hyperintense on T2-weighted MRI and hypointense on computed tomography scans, commonly referred to as White Matter Lesions (WMLs). MRI features of WMLs are correlated with dilatation of perivascular spaces, especially in the frontal, and parietal subcortical area, and around the ventricular system. These lesions are often seen in elderly people and are related to various geriatric disorders, such as cerebrovascular disease, cardiovascular disease. The literature extensively reports the association between WMLs and dementia [6], dizziness [7] and psychiatric disorders, such as major depressive disorder, and bipolar disorder [8]. The aim of our work was to investigate the correlation WMLs and presbyvestibulopathy. The elderly patients with mild bilateral peripheral vestibular hypofunction, without a clear diagnosis and despite clinical and vestibular assessment, should undergo brain imaging to assess the level of WML in particular is to focus scientific attention on aspects of cognitive–vestibular. Ageing and patient comorbidities and multiple vascular risk factors are important facilitating factor could contribute to the onset of a vestibular syndrome. Vestibular disorders and cognitive function such as anxiety, fear, phobia and depression are intimately related and this relationship could be explained by the fact that they have some common neural pathways.
Materials & methods
The authors retrospectively analyzed patients who referred to the LAB of Clinical and Instrumental Vestibology, Poliambulanza Foundation Hospital in Brescia between August 2022 and January 2023. A total of 48 patients (24 females, 24 males; average age males: 71 years; average age females: 72 years; age range: 57-87 years) were enrolled in the present study. Vascular risk factors, such as heart disease, hypertension, diabetes, hypercholesterolemia, and previous stroke, were evaluated for each patient. All subjects had an otoneurological examination including the observation of eye movements (spontaneous, gaze evoked, and positional nystagmus). For the PVP instrumental diagnostic criteria, only values from the Video-Head Impulse Test (v-HIT) were selected, without considering the rotary chair, or caloric testing, given that the Vestibular Ocular Reflex (VOR) gain quantification is mainly possible with the former. Since there is no agreement in the literature about upper threshold of VOR gain to define a mild vestibulopathy, we decided to establish a VOR gain threshold as <0.95 and >0.6. In fact, the ICVD definition of BVP defined a VOR gain <0.6 bilaterally as one of the diagnostic criteria. The lower VOR gain threshold for PVP, which defines a mild vestibulopathy, was therefore set at 0.6. An upper limit of 0.8 was selected as the upper bound by synthesizing data across several studies. One study [9] assessed VOR gain in a normative sample of 62 adults and observed that the lower limit of normal horizontal VOR gain (2SD below the mean) was 0.79 at 80 ms and 0.75 at 60 ms. However, another study [10] of 243 healthy older adults revealed that a VOR gain value of <0.9 was associated with a significantly higher prevalence of compensatory saccades, which are considered a marker of VOR deficiency. Future revisions of these criteria should consider whether the cutoffs for mild vestibular impairment in older adults should be refined.
Participants were evaluated following a one-month period without the use of sedative medications (e.g., benzodiazepines), which are commonly prescribed to older individuals. All participants underwent 1,5 tesla MRI scans. The MRI protocol consisted of axial T1-weighted spin-echo images, axial, dual T2weighted Turbo spin-echo images and coronal fluid-attenuated inversion (FLAIR) images. For each image series, 20 slices covering the entire brain were obtained. White Matter hyperintensities were rated according to the Fazekas scale [11]. This is a scale of WMLs burden according to the lesions load (0: none or a single punctate lesion; 1: multiple punctate lesions=mild; 2: early confluence of lesions=moderate; 3: large confluent lesions=severe). WMLs were evaluated by lobe location, number and size. Automated quantification methods of to assess WMLs are rapidly evolving. This classification was proposed by Fazekas et al. in 1987 and at the time of writing (late 2016) it remains the most widely used system for describing white matter disease severity in publications. It is not generally used in clinical practice, with terms such as ‘mild’, ‘moderate’ and ‘severe’ being favored. Therefore, and should make it is easier to apply fine quantitative criteria for the sub-classification WMLs based on the anatomical and pathological criteria of WMLs, that comprises five areas, which supported a core region hypothesis in the central vestibular circuit: a) Subcortical area: prefrontal cortex frontal lobe, premotor cortex, frontal eye fields; b) deep periventricular; c) basal ganglia semioval center, corona radiata, caudate nucleus, globus pallidus, lentiform nucleus, thalamus, external capsule; d) parieto-insular vestibular cortex , retroinsular cortex, posterior insula, inferior parietal lobule, intraparietal sulcus, central sulcus; e) limbic system, cingulate anterior dorsal, hippocampus, amygdala, ipothalamic.
Results
All enrolled patients matched to primary factors that describe presbyvestibulopathy of the Consensus document of the classification committee of the Bárány Society: chronic vestibular syndrome (at least 3 months duration), gait disturbance, chronic dizziness, mild bilateral peripheral vestibular hypofunction documented by VOR gain measured by video-HIT between 0.6 and 0.95 bilaterally and age ≥60 years. In our study, 48 patients with complex visual stimuli, abnormalities on MRI imaging were found. The Table 1 shows the diagnostic criteria Consensus document of the classification committee of the Bárány Society of Presbyvestibulopathy (2019).
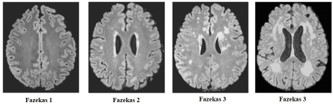
V-HIT results are outlined in Table 2. The estimates of peak location result from analysis of several neuroimaging of the WMLs, which can be subdivided in five major areas: a) subcortical area: frontal lobe, prefrontal cortex (34,6%); b) deep periventricular (34,6%); c) parieto-insular vestibular cortex (22,4%); d) basal ganglia: semioval center (22,4%), corona radiata (20,4%), caudate nucleus (6,1%), putamen (6,1%), globus pallidus (6,1%); e) thalamus (8,1%). By classification, we aim to make WMLs finding more relevant functionally. The Tab. III shows the classification and correlation between WMLs and central vestibular circuit. The Tab. IV, shows the Fazekas-scale provides an overall impression of the presence of WMH in the entire brain. On MRI, white matter hyperintensities (WMH) and lacunes both of which are frequently observed in the elderly are generally viewed as evidence of small vessel disease. It is best scored on transverse FLAIR or T2-weighted images (Figure 1). As expected, age was strongly associated with WMLs. Furthermore, sex, arterial hypertension, and diabetes were significantly associated with Fazekas scale (see Table 4. Demographic and clinical characteristics of the group as defined by amount of white matter hyperintensities.), which was not the case for hypercholesterolemia. The Table 5 illustrates the clinical characteristic of prespyvestibulopathy symptoms associated with Fazekas scale: postural imbalance (91,6%) and gait disturbance (83,3%) are associated with Fazekas 1 (focal lesion), while the chronic dizziness (79%) are associated Fazekas 2 (early confluent), finally the recurrent falls (10,4%) associated with Fazekas 3 (widespread changes in white matter). The data were also correlated of the v-HIT test with Fazekas scale: 41,6% (Fazekas 1), 47,9% (Fazekas 2) and 10,4% (Fazekas 3). Performance in v-HIT and results of the statistical analysis post hoc comparisons are illustrated in Tab. V. Fazekas groups 1 significantly in postural imbalance (91,6%) and gait disturbance (83,3%), while chronic dizziness (79%) is associated with Fazekas 2. Interestingly, only 5 patients had recurrent falls, all are related to the Fazekas 3 group.
Table 1: Diagnostic criteria - Consensus document of the classification committee of the Bárány Society of Presbyvestibulopathy (2019).
| Primary factors that describe presbyvestibulopathy: A. Chronic vestibular syndrome (at least 3 months duration) with at least 2 of the following symptoms: 1. Postural imbalance or unsteadiness; 2. Gait disturbance; 3. Chronic dizziness; 4. Recurrent falls. B. Mild bilateral peripheral vestibular hypofunction documented by at least 1 of the following: 1. VOR gain measured by video-HIT between 0.6 and 0.83 bilaterally; 2. VOR gain between 0.1 and 0.3 upon sinusoidal stimulation on a rotatory chair (0.1 Hz, Vmax = 50–60°/sec); 3. Reduced caloric response (sum of bithermal maximum peak SPV on each side between 6 and 25°/sec); C. Age≥60 years; D. Not better accounted for by another disease or disorder. |
Table 2: Vestibular Ocular Reflex (VOR) gain measured by video-head impulse test (v-HIT): gain thresholds to >0,6 and <0,95.
| N° | Female | Left lateral semicircularcanal | Right lateral semicircular canal | N° | Male | Left lateral semicircular canal | Right lateral semicircular canal |
|---|---|---|---|---|---|---|---|
| 1 | f | 0,63 | 0,7 | 1 | m | 0,77 | 0,85 |
| 2 | f | 0,82 | 0,68 | 2 | m | 0,88 | 0,78 |
| 3 | f | 0,93 | 0,84 | 3 | m | 0,66 | 0,77 |
| 4 | f | 0.87 | 0,91 | 4 | m | 0.84 | 0,88 |
| 5 | f | 0,91 | 0,67 | 5 | m | 0,78 | 0,84 |
| 6 | f | 0,71 | 0,95 | 6 | m | 0,79 | 0,85 |
| 7 | f | 0,76 | 0,84 | 7 | m | 0,82 | 0,86 |
| 8 | f | 0,70 | 0,93 | 8 | m | 0,78 | 0,84 |
| 9 | f | 0,83 | 0,95 | 9 | m | 0,71 | 0,78 |
| 10 | f | 0,91 | 0,85 | 10 | m | 0,93 | 0,85 |
| 11 | f | 0,84 | 0,92 | 11 | m | 0,78 | 0,65 |
| 12 | f | 0,91 | 0,66 | 12 | m | 0,78 | 0,84 |
| 13 | f | 0,74 | 0,67 | 13 | m | 0,78 | 0,89 |
| 14 | f | 0,80 | 0.93 | 14 | m | 0,77 | 0.76 |
| 15 | f | 0,84 | 0.92 | 15 | m | 0,71 | 0.76 |
| 16 | f | 0,91 | 0,66 | 16 | m | 0,78 | 0,66 |
| 17 | f | 0,84 | 0,98 | 17 | m | 0,85 | 0,91 |
| 18 | f | 0,74 | 0,67 | 18 | m | 0,82 | 0,78 |
| 19 | f | 0,80 | 0,93 | 19 | m | 0,93 | 0,80 |
| 20 | f | 0,85 | 0,93 | 20 | m | 0,76 | 0,81 |
| 21 | f | 0,90 | 0,85 | 21 | m | 0,82 | 0,78 |
| 22 | f | 0,65 | 0,94 | 22 | m | 0,69 | 0,75 |
| 23 | f | 0,68 | 0,95 | 23 | m | 0,85 | 0,93 |
| 24 | f | 0,61 | 0,89 | 24 | m | 0,72 | 0,85 |
Table 3: Anatomical area distribution of small White Matter Lesion (WMLs) and classification on the four stage Fazekas scale.
| Anatomical area | Number WMLs (%)* | Fazekas Scale** |
|---|---|---|
| 1. Subcortical area (juxtacortical: within 4 mm from corticomedullary junction) | ||
| Frontal lobe PreFrontal Cortex (PFC) |
17(35%) | 1:7 pts 2:10 pts 3:0 pts |
| 2. Deep periventricular (3-13 mm from ventricular surface) | ||
| Periventricular area | 17(35%) | 1:8 pts 2:8 pts 3:1 pts |
| Semioval Center | 11(23%) | 1:6 pts 2:5 pts 3:0 pts |
| Corona Radiata | 10(21%) | 1:5 pts 2:5 pts 3:0 pts |
| 3. Basal ganglia (juxtaventricular within 3 mm from ventricular surface) | ||
| Internal capsule: caudate Nucleus (Neostriatum); Globus pallidus (Paleostriatum); Lentiform nucleus (Putamem+Globus Pallidus); Thalamus | 10(21%) | 1:3 pts 2:3 pts 3:4 pts |
| External capsule: pallido-thalamic tracts lenticular fasciculus | 4(8%) | 1:4 pts |
| 4. Parieto-Insular Cortex (PIC) | ||
| Retroinsular Cortex, Posterior Insula, Inferior Parietal Lobule, Intraparietal central sulcus | 11(23%) | 1:0 pts 2:11 pts 3:0 pts |
| 5. Limbic system | ||
| Cingulate Anterior Dorsal (CgAd), Hippocampus, Amygdala (Archistriatum), Ipothalamic. | 5(10,4%) | 1:2 pts 2:2 pts 3:1 pts |
*The total number of small White Matter Lesion is greater than the number of patients (48 pts) because one patient can have more than one lesion. **Classification of white matter hyperintensities on the four stage Fazekas scale: The healthy brain (Fazekas 0), contrasted with punctiform (Fazekas 1), early confluent (Fazekas 2), and diffuse confluent (Fazekas 3).
Table 4: Demographic and clinical characteristics of the group as defined by amount of white matter hyperintensities.
| Fazekas scale | ||||
|---|---|---|---|---|
| 1 | 2 | 3 | Group comparisons | |
| N(%) | 21(43,7) | 45(93,7) | 2(4,1) | |
| Sex (%male) | 50 | 51,8 | 31,5 | P<0,05* |
| Diabetes | 13,5 | 16 | 24 | P<0,01** |
| Arterial hypertension | 59,5 | 83,5 | 85,5 | P<0,001*** |
| Hypercolesterolemia | 35,5 | 40.5 | 36,8 | ns |
*p<0,05, **p<0,01, ***p<0,001 as tested with univariate ANOVA or Chi-square statistic.
Table 5: Clinical characteristic of the symptoms of prespyvestibulopathy associated with Fazekas scale.
| Fazekas 1 | Fazekas 2 | Fazekas 3 | |
|---|---|---|---|
| Postural imbalance | 44(91,6%) | ||
| Gait disturbance | 40(83,3) | ||
| Chronic dizziness | 38%(79%) | ||
| Recurrent Falls | 20(41,6%) | 5(10,4%) | |
| v-HIT | 20(41,6%) | 23(47,9%) | 5(10,4%) |
Discussion
We systematically studied dizzy patients with MRI to map WMLs and to verify the correlation between WML and presbyvestibulopathy. The comorbidities also play an important role in aggravating the physiological degeneration of the balance system, in our study, the most important vascular risk factor are arterial hypetension (79%), hypercholesterolemia (55%), cardiac disease (35%), diabetes mellitus (31%) and cerebrovascular events (15%). Several vascular risk factors could contribute to the onset of a vestibular syndrome called vascular vertigo due to its vascular etiology. In our study, 48 patients with presbyvestibulopathy on MRI imaging were found WMLs in prefrontal cortex (34,6%) and deep periventricular (34,6%). The estimates of peak location result from analysis of several neuroimaging of the WMLs, which can be subdivided in two areas: prefrontal cortex (34,6%) and deep periventricular (34,6%). The total number of small White Matter Lesion is greater than the number of patients (48 pts) because one patient can have more than one lesion, the areas affected by the WMLs are different, there is no single area. The characteristic vestibular disorders are postural imbalance, correlated with prefrontal cortex, and mild bilateral peripheral vestibular hypofunction (VOR gain measured by video-HIT between 0.6 and 0.83 bilaterally), which is correlated with WMLs located in deep periventricular area and recurrent falls are documented in both prefrontal cortex and periventricular cortex area. In older adults, alteration in behavior, including changes in balance control and falls, have been associated with the term “fear of falling” and with restricted activity, decreased independence quality of life, anxiety are evidence [12]. Patients with spatial working deficit, anxiety and depression are correlated with WMLs located in the Parieto-Insular Cortex (PIC), which includes, retro insular cortex, posterior insula, inferior parietal lobule and intraparietal central sulcus (22,4%). Other cognitive symptoms, such as fear, were found to correlate with white WMLs located in the semioval center (22.4%), while stress correlated with WMLs located in the corona radiata (20.4%). We hypothesize that it is not the etiology of WMLs but rather the distribution of their multifocal, prefrontal cortex, parieto-insular cortex, limbic structures, thalamus, hippocampal, cerebellar and cerebral cortex structures that may account for the symptoms of presbyvestibulopathy, particularly in relation to the association between dizziness and negative emotions. There is evidence that periventricular white matter lesions are particularly related to cognitive decline, whereas subcortical white matter lesions may be related to late onset depression. WMLs are extremely common in elderly people and it is clear that the small and often occult cerebrovascular lesions that we label as ‘‘leukoaraiosis’’ or ‘‘WMLs’’ are not clinically ‘‘silent’’ as once believed. The association between gait and balance abnormalities and WMLs was further strengthened by several prospective follow-up studies. Baloh et al. [12] followed 59 normal elderly participants aged 75 years and older for a minimum of 8 years (range, 8–10 years). They found that WMLs were highly correlated with changes in gait and balance. Direct evidence for an association between WMLs and falls is limited, but it can be inferred from the multitude of studies showing associations between WML and balance disturbances [13,14]. WMLs may interrupt frontal lobe circuits responsible for normal gait and balance or they may interfere with long loop reflexes mediated by deep white matter sensory and motor tracts. In addition, the periventricular and subcortical distribution of WMLs could interrupt the descending motor fibers arising from medial cortical areas, which are important for lower extremity motor control. WMLs of older people with depression are most prominent in frontal subcortical areas, and thus basal ganglia and their frontal and limbic connections are affected. Common geriatric syndromes such as cognitive impairment, gait disturbance, depression, and urinary incontinence have been shown to be closely associated with WMLs. Patient with presbystasia, are susceptible to visual motion, esacerbation of dizziness walking and watching moving scenes. The study reveals widespread vestibular activations in the motor, visual, and somatosensory cortex, associative parietal cortex, prefrontal cortex, thalamus and limbic structures. Finally, temporal integration and its ancillary prefrontal functions (attention, working memory, set, monitoring) operate within the broader dynamics of the perceptionaction cycle, which is founded on basic biological principles. The traditional classification of vestibular disorders is based on the anatomical site of the lesion and distinguishes between the peripheral and the central vestibular system. This classification could not reflect the clinical syndrome, which may involve both the peripheral and the central vestibular system, leading to “higher vestibular function” disorders, since inputs from visual, vestibular and proprioceptive systems are integrated and elaborated through cognitive functions [15]. Additionally, a central lesion, such as cerebellar infarctions, may mimic a peripheral disorder and could be misinterpreted [16]. Additionally, a recent retrospective case analysis showed increased severity and frequency of WML in subjects with unexplained dizziness, suggesting that WML could contribute to the development of dizziness. With increasing age, comorbidities, a progressive loss of function of the vestibular, visual and proprioceptive systems can lead to balance problems. It has not yet been demonstrated but it is likely that rotational vertigo is a type of visual input with a component of emotional distress and fear that can activate the amygdala and related circuits of memory [17]. Patients with persistent non-vertiginous dizziness that may be associated with one or more of the following descriptors: light-headedness, heavy-headedness, unsteadiness ambulating, spinning sensation, in the head with no motion of the visual field, rocking sensation, chronic hypersentivity to self-motion. Symptoms are most severe when the patient is standing and walking and less severe when sitting or lying down [18-20]. Vestibular system lesions (vestibular cortical relationships, vestibular-thalamic relationships, vestibular-limbic relationships) produce a disruption in top kinetic memory disorganization, spatial working memory deficit, anxiety and depression, saccadic visual suppression and other cognitive symptoms: fear, panic and stress [20-22]. The idea is that all these areas, including the prefrontal area, process ego motion information and may be involved in planning motor synergies to counteract loss of equilibrium [23,24]. Studies into the neural mechanism underlying these interactions have focused on stress-induced synaptic plasticity impairments in the hippocampus [25,26]. A further limitation is the crosssectional design of this study, which makes it difficult to draw conclusions whether depicted changes are consequence of the disorders or whether pre-existent or acquired alterations of these regions make patients more susceptible to developing presbyvestibulopathy [27,28]. Although an analysis without three patients suffering from comorbid anxiety disorder did not alter results, anxiety disorder is a well-known psychiatric comorbidity which has a higher prevalence than depression in presbyvestibulopathy [29,30]. We have to acknowledge that in two thirds we could not identify a specific cause for presbyvestibulopathy because information regarding non vestibular triggering events was not systematically evaluated. Our data provide new insights into the pathophysiology of presbyvestibulopathy. The detected chronic vestibular symptoms in regard to maladaptation and modulation mechanism reflecting cortical plasticity of the multisensory vestibular system over time.
Definitely, RM Angiography is the gold standard to reveal all eventual stenosis but the purpose of this work is to correlate cerebral vessel small disease with presbyvestibulopathy.
Although this study was carefully conducted, some critical remarks must be mentioned. Quantification of white matter hyperintensities may be interpreted as conservative approach, as WMLs can potentially be extracted with the help of automated computer algorithms, and no spatial information about the lesion was considered. We used the Fazekas scale, as WMLs classification is simple, reliable and highly correlated with volumetric measures of WMLs. Another limitation refers to the small number of individuals classifies as Fazekas.
Conclusion
Our data reveal the functional importance of the prefrontal cortex in modulating anxiety and other emotional behaviors through its connections to amygdala. Numerous neuronal pathways link the vestibular network with limbic and hippocampal structures. In recent years, many authors have proposed the existence of a “vestibular cortex” [31,32]. We propose the vestibular cognitive map, a growing literature on the physiology of the vestibular system has demonstrated the existence of projections from the vestibular nuclei to the cerebral cortex. The goal of this review is to focus scientific attention on the aspects of cognitive–vestibular interactions indicated by a wide range of results in the literature. Ageing and patient comorbidities and multiple vascular risk factors are important facilitating factor could contribute to the onset of a vestibular syndrome. Vestibular disorders and cognitive function (anxiety, fear, phobia and depression) are intimately related and this relationship could be explained by the fact that they have some common neural pathways [33]. The correlation could be further supported by the high prevalence of frontal and parieto-insular cortex of WMLs among people with presbyvestibulopathy [34]. Our findings suggest that elderly patients with persistent non-vertiginous dizziness, without a clear diagnosis and despite clinical and vestibular assessment, should undergo brain imaging to assess the level of WML [35]. Our personal experience with patients suffering from vertigo confirmed these correlations between WML and cognitive functions and evaluate the possibility to employ a pharmacological therapy with the purpose to avoid the chronic phase of the anxiety and fear, symptoms in these clinically complex patients [36]. Presbyvestibulopathy is the result of age-related physiological changes in the three sensory systems and their central connections that contribute to balance. In all likelihood, presbyvestibulopathy is a complex condition involving many intertwined systems rather than a lesion within the vestibular system only, thus evaluation and management need to be holistic. Balance problems can have detrimental consequences and are associated with falls, loss of quality of life and psychological sequelae such as anxiety, depression and panic. Healthcare practitioners need increased awareness of the pathophysiology of presbyvestibulopathy and its possible impact.
Declarations
Conflict of interest: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-forprofit sectors.
Author contributions: PG: conceptualization, methodology; GL, LK, CB, LC: data collection, writing – original draft; PG, AD, PU: formal analysis, writing – review & editing the final manuscript.
Ethical consideration: All patients underwent routinelyperformed tests only, without invasive or experimental procedures. Informed consent was obtained from all participants and the study was performed in accordance with the Declaration of Helsinki. The study was conducted according to the criteria set by the declaration of Helsinki and each subject signed an informed consent before participating to the study.
References
- Agrawal Y, Van de Berg R, Wuyts F, et al. Magnusson M, Oh E, Sharpe M, Strupp M. Presbyvestibulopathy: Diagnostic criteria Consensus document of the classification committee of the Bárány Society. Journal of Vestibular Research. 2019; 29: 161-170.
- Maarsinggh OR, Dros J, vad der Windt DA, et al. Diagnostic Indicators of Anxiety and Depression in Older Dizzy Patients in Primary Care. J Ger Pschi Neurol. 2011; 24: 98-107.
- Gamba P. Vestibular-limbic relationships: Brain mapping. Insights Depress Anxiety. 2018; 2: 7-13.
- Ahmad H, Cerchiai N, Mancuso M, et al. Are white matter abnormalities associated with “unexplained dizziness”?. J Neurol Sci. 2015; 358: 428-31.
- Colledge N, Lewis S, Mead G, el al. Magnetic resonance brain imaging in people with dizziness: a comparison with non-dizzy people. J Neurol Neurosurg Psychiatry. 2002; 72: 587-89.
- Staab JP. Chronic dizziness: the interface between psychiatry and neuro-otology. Curr Opin Neurol. 2006; 19: 41-48.
- Guidetti G. The role of cognitive processes in vestibular disorders. Hearing, Balance Commun. 2013; 11: 3-35.
- Ruckenstein MJ, Staab JP. Chronic Subjective Dizziness. Otolaryngol Clin N Am. 2009; 42: 71-77.
- Harvood RH. Visual problems and falls. Age Ageing. 2001; 30: 13-18.
- Mener DJ Betz J, Genther D, Chen, Lin FR. Hearing loss and depression in older adults. J Am Geriatr Soc. 2013; 6: 1627.
- Fazekas F, Chawluk JB, Alavi A, et al. MR signal abnormalities 1,5 T in Alzheimer’s dementia and normal aging. Am J Roentgenol. 1987; 149: 351-56.
- Baloh RW, Yue Q, Socotch TM, et al. White matter lesions and disequilibrium in older people. I. Case-control comparison. Arch Neurol. 1995; 52: 970-74.
- GE Hicks, M Shardell, DE Alley, RR, et al. Absolute strength and loss of strength as predictors of mobility decline in older adults: The InCHIANTI study. J Gerontol A Biol Sci Med Sci. 2012; 67: 66–73.
- G. Ishiyama, Imbalance and vertigo: The aging human vestibular periphery. Semin Neurol. 2009; 29: 491–499.
- DJ Genther, J Betz, S Pratt, et al. Association of hearing impairment and mortality in older adults. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences. 2014; 70: 85–90.
- NS Gittings, JL Fozard. Age related changes in visual acuity. Exp Gerontol. 1986; 21: 423–33.
- Belal A, Glorig A. Dysequilibrium of Ageing (Presbyastasis). J Laryngol Otol. 1986; 100: 1037–41.
- Herdman S, Blatt P, Schubert M, et al. Falls in patients with vestibular deficits. Am J Otolaryngol. 2000; 21: 847–51.
- Staab JP, Ruckenstein MJ. Which comes first? Psychogenic dizziness vs. otogenic anxiety. Laryngoscope. 2003; 113: 1714-18.
- Gamba P, Pavia M. White matter lesions and vascular vertigo: clinical correlation and findings on cranial magnetic resonance imaging. Eur Rev for Med Pharmacol Sci. 2016; 20: 2786-91.
- Kuo HK, Lipsitz A. Cerebral White Matter Changes and geriatric Syndromes: Is There a Link?. Journal of Gerontology. 2004; 8: 818-26.
- Pollack L, Osherov M, Berkovitz, et al. Magnetic resonance brain imaging in patients with visual vertigo. Brain an Behavior. 2015; 5: e00402.
- Brandt T, Strupp M, Dieterich M. Towards a concept of disorders of “higher vestibular function”. Front Integr Neurosci. 2014; 8: 47.
- Dieterich M, Brandt T. The bilateral central vestibular system: its pathways, functions, and disorders. Ann NY Acad Sci. 2015; 1343: 10-26.
- Matsusue E, Sugihara S, Fujii S, et al. White matter changes in elderly people: MR-pathologic correlations. Magn Reson Med Sci. 2006; 5: 99-104.
- Debette S, Markus HS. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. Br Med J. 2010; 341: c3666.
- McGaugh JL. The amygdala modulates the consolidation of memories of emotionally arousing experiences. Annu Rev Neurosci. 2004; 27: 1-28.
- Zu Eulenburg P, Caspers S, Roski C, el al. Meta-analytical definition and functional connectivity of the human vestibular cortex. Neuro Image. 2012; 60: 162-69.
- Mazzola L, Lopez C, Faillenot I, et al. Vestibular responses to direct stimulation of the human insular cortex. Ann Neurol. 2014; 76: 609-19.
- Teggi R. Dizziness anxiety and migraine. In: Colombo B, Teggi R, editors. Vestibular migraine and related symptoms. Springer International Publishing, Switzerland. 2014: 159-173.
- Gufoni M, Guidetti G, Nuti D, et al. The relationship between cognitive impairment, anxiety-depression symptoms and balance and spatial orientation complaints in the elderly. Acta Otorhinolaryngol Ital. 2005; 25: 12-21.
- Teggi R, Caldirola D, Perna G, et al. Vestibular testing in patient with panic disorders and chronic dizziness. Acta Otorhinolaryngol Ital. 2007; 27: 243-47.
- Brandt T, Dieterich M. The vestibular cortex. Its locations, functions, and disorders. Ann NY Acad Sci. 1999; 871: 293-312.
- Perna G, Alpini D, Caldirola D, et al. Panic disorder: the role of the balance system. J Psychiatr Res. 2001; 35: 279-86.
- Simon NM, Pollack MH, Tuby KS, et al. Dizziness and panic disorders: a review of the association between vestibular dysfunction and anxiety. Ann Clin Psychiatry. 1998; 10: 75-80.
- Kirsch V, Keeser D, Hergenroeder T, et al. Structural and functional connectivity mapping of the vestibular circuitry from human brainstem to cortex. Brain Struct Funct. 2015; 3: 12911308.