Volume 1, Issue 2
Article Type: Research Article
Combination of 2,4 dinitrophenol and tempol mitigates synaptic and non-synaptic mitochondria and neurobehavioral outcomes post-anoxia
Puneet K Samaiya1,2; Santosh Kumar Prajapati1,3; Ashok Kumar4; Sairam Krishnamurthy1*
1Neurotherapeutics Laboratory, Pharmaceutical Engineering and Technology, Indian Institute of Technology (Banaras Hindu University), India.
2Department of Pharmacy, Shri G.S. Institute of Technology & Science, India.
3Department of Neurosurgery and Brain Repair, University of South Florida, USA.
4Department of Pediatrics, Institute of Medical Sciences, Banaras Hindu University, India.
*Corresponding author: Sairam Krishnamurthy, PhD
Professor in Pharmacology, Department of Pharmaceutical
Engineering and Technology, Indian Institute of Technology (Banaras Hindu University), Varanasi-221005, India.
Email ID: saibliss@hotmail.com & ksairam.phe@iitbhu.ac.in;
Tel: +91-9935509199, Fax: +91-542-2368428
Received: Sep 06, 2025
Accepted: Oct 09, 2025
Published Online: Oct 16, 2025
Journal: Annals of Gerontology and Geriatrics
Copyright: Krishnamurthy S et al. © All rights are reserved
Citation: Samaiya PK, Prajapati SK, Kumar A, Krishnamurthy S. Combination of 2,4 dinitrophenol and tempol mitigates synaptic and non-synaptic mitochondria and neurobehavioral outcomes post-anoxia. Ann Gerontol Geriatr Res. 2025; 1(2): 1023
Abstract
Neonatal anoxia is a global neuropathological condition that originated due to the severe deprivation of oxygen in the developing brain during the time of birth. We have recently reported the progressive loss of mitochondrial function from d-1 to d-7 after anoxia. This study is the first to investigate the effects of combination of 2,4-dinitrophenol (2,4-DNP)+Tempol (D+T) on both synaptosomal (neuronal) and non-synaptosomal (glial and neuronal soma)-derived mitochondria seven days after the second anoxic episode in rat pups. Studies have shown that mitochondrial Ca2+ overload and ROS are two major determinants in mitochondrial functioning and the two populations differ in their Ca2+ handling capacity. We observed (D+T) combination significant attenuates in both mitochondrial populations in terms of various biochemical (s-3,s-4,RCR, LPO, NO, SOD,CAT, Ca2+, MPT) and molecular (cytochrome-C and caspase-9/3) parameters compared to anoxia group. However, we observed no marked synergistic effect in combination D+T as compared to their individual effects.
Keywords: Ca2+ overload; Synaptic mitochondria; Non-synaptic mitochondria; Cytochrome-C; Caspase-9/3.
Introduction
Neonatal anoxia is a common obstetric complication resulting due to lack of oxygen supply to the fetus or newborn to a small period at the time of birth [7]. India’s under-five mortality rate is now on par with the global average of 39 deaths per 1,000 live births. Of these deaths, more than 80% are due to perinatal asphyxia, a condition that occurs during childbirth and can lead to breathing problems in newborns [37]. Currently, there are no FDA-approved drugs that can prevent or reverse the neurological damage caused by anoxia. Therapeutic hypothermia is a standard treatment that can improve the chances of recovery, but it is partially effective. In our earlier studies we performed a temporal profiling of mitochondrial dysfunction that progresses from primary to secondary insult [36,38] after anoxia. This helped us to identify the time point of intervention (2,4 DNP and tempol) that were administered within 5 minutes of second episode of anoxia exposure to prevent further neuronal damage through preserving mitochondrial bioenergetics [39,40]. Soon after anoxia there is an occurance of excitotoxicity, which in turn increases Ca2+ overload and increase Reactive Oxygen Species (ROS) production, inhibiting Adenosine Triphosphate (ATP) synthesis, impair Oxidative Phosphorylation (OXPHOS), as well as enhanced Mitochondrial Permeability Transition Pore (mPTP) formation [37]. Previous reports have shown that by reducing Mitochondrial Membrane Potential (MMP), their uptake of Ca2+ can be reduced [23,27]. Our findings confirms that mitochondria can be a promising therapeutic target for neuroprotection after anoxic injury. Although, these studies were performed in crude (synaptic and non-synaptic) mitochondria within the cortex. However, crude mitochondrial preparations can facade the effect of injury on synaptic mitochondria due to the high glia to neuron ratio within the cortex [19]. The isolated mitochondria within the brain (cerebral cortex) can be categorized into two separate groups: one is a synaptic population comprising mitochondria from both preand post-synaptic neurons [29], while the other is a non-synaptic population consisting of mitochondria originating from various cell types including neurons (axonal, dendritic, somal) and non-neuronal cells (glial, endothelial, etc.) [20]. Previous studies with different neuronal injury models have reported that synaptic mitochondria are more vulnerable to neuronal injury as compared to non-synaptic mitochondria. Further, these studies suggested that the total mitochondrial preparations can mask the effect of injury on synaptic mitochondria due to the high glia to neuron ratio within the cortex [3,19,20]. Similarly some studies have shown that the synaptic mitochondria are more susceptible to Ca2+-induced MPT threshold and Reactive Oxygen Species (ROS) production as compared to non-synaptic mitochondria [3,26]. However, the major contribution of a specific mitochondrial population (synaptic or non-synaptic) is a matter of study in neonates post anoxic injury. Apart from this to evaluate the variations in Ca2+ handling capacity and permeability pore opening in different mitochondrial fractions and the treatment strategies which are selective to a particular population of mitochondria to improve their function are also not elucidated in anoxia. Furthermore, anoxia-induced mitochondrial dysfunction has been implemented as a prime cause of acute sensorimotor deficits [36,38]. However, using different models, previous studies have considered anoxia to be a prime cause of long-term neurodevelopmental delays [4,12,41,47]. Moreover, a continuous development of Hypothalamic–Pituitary–Adrenal (HPA) axis is crucial for the proper function of the organism. Unluckily, the perinatal events alter the subsequent phenotypical response to stress by changing the set-point of the HPA axis [8,33]. As a result, an alteration in corticosterone levels takes place [34]. Formerly, in our recent unpublished study, it has been demonstrated that treatment with uncoupler 2,4 DNP or antioxidant tempol within 5 minutes of second anoxic episode causes improvement in mitochondrial function as well as in sensorimotor performance in neonates. However, the long-term effect of anoxia on neurobehavior changes are yet to be elucidated.
Therefore, the purpose of the present study is to identify the significant changes in bioenergetics between synaptic and nonsynaptic mitochondria post anoxic injury. Further, to compare the ability of isolated synaptic versus nonsynaptic cortical brain mitochondria regarding Ca2+ handling and other biochemical and biomolecular studies on rat pups (P10) after anoxia. Furthermore to study the combined effect of 2,4 DNP and tempol on the basis of our previous studies [39,40] for any synergistic effect in the two mitochondrial populations in terms of mitochondrial respiration, (s-3,s-4 and RCR), oxidative stress, antioxidant enzyme, maintaining Ca2+ homeostasis, pro-apoptotic Bcl-2 family proteins and apoptotic proteins (cytochrome-C, caspase-9/3). Further we observed the effect of combination in long term (d-21 up to d-150) neurobehavioral outcome like changes in spontaneous locomotor activity as a measure of hyperactivity, spatial recognition memory impairments as a measure of cognitive impairments and further anxiety and depression-like behavior and further plasma corticosterone on d-150 after anoxia.
Materials & methods
Animals
The Institute of Medical Sciences, Banaras Hindu University (IMS-BHU) Central Animal House provided the 180–220 g pregnant Charles Foster albino rats. The animals were kept in polypropylene cages with controlled lighting and dark cycles, with a temperature of 25±1°C and a relative humidity of 45–55%. A birth litter count of 8–12 pups per rat, weighing 6–8 g, was employed. The rats were roughly 30 hours old. The BHU Institutional Animal Ethical Committee accepted the experimental protocols (Protocol No. Dean/11-12/CAEC/328). The National Research Council US Committee for the Update of the Guide for the Care and Use of Laboratory Animals (2011) criteria for laboratory animal care were followed in conducting all of the experiments.
Anoxia model
As previously specified and validated, the anoxic technique was executed [36]. In brief, subjects who were about 30 hours old were put into a non-hermetic chamber (for causing anoxia) with a flow rate of 3 L/min and a pressure of roughly 101.7 of 100% nitrogen (N2) for two 10-minute anoxic episodes spaced 24 hours apart. To prevent hypothermia, the chamber was partially submerged in warm water (between 35 and 37°C). The control group underwent the same experimentation, except the nitrogen in the chamber was replaced with air. The animals were randomly returned to the dams after exposure, and on day seven, they were slaughtered. After the brains were quickly removed, the cortical section was utilized right away for western blot and mitochondrial isolation. Studies on mitochondrial respiration were carried out as soon as the mitochondria were isolated. Additionally, mitochondrial pellets were kept for additional biochemical assessment at -80°C.
Drug preparation and dosing
From the earlier research, the lowest effective doses of tempol (75 mg/kg) and 2,4 DNP (2.5 mg/kg) were employed [39]. Prior to intraperitoneal administration, 2, 4 DNP (Himedia) and tempol were newly produced in DMSO (i.p.). Six groups (n=6) of six rats each were created by random selection: (1) Control; (2) Anoxia; (3) DNP 2.5 mg/kg; (4) Tempol 75 mg/kg; and (5) DNP+Tempol. As previously mentioned, the medications were given within five minutes of the second anoxia episode.
Chemicals
Sodium pyruvate, malate, ADP, succinate, oligomycin, carbonyl cyanide 4-(trifluoromethoxy) phenylhydrazone (FCCP), Rotenone, tetramethylrhodamine methyl ester (TMRM) and Griess reagent were procured from Sigma-Aldrich (St. Louis, MO). The Elisa test for caspase-3(elabscience, USA, Cat# E-ELR0160) caspase-9 (elabscience, USA, Cat# E-EL-R0163) and cytochrome-c (elabscience, USA, Cat# E-EL-R0006) procured from Elabsciences USA.
Synaptic and non-synaptic mitochondria isolation
With a few minor adjustments, mitochondrial isolation was carried out in accordance with earlier instructions [13,36]. After adding 4 mL of isolation buffer with EGTA to an all-glass Dounce homogenizer, which contained 215 mM mannitol, 75 mM sucrose, 0.1% w/v bovine serum albumin, 20 mM HEPES buffer, and 1 mM of EGTA in 100 ml of distilled water with pH adjusted to 7.2 with KOH, the rat cortex was centrifuged for 5 minutes at 4°C at 1300×g. To obtain a more compact mitochondrial pellet, each supernatant was topped up with isolation buffer containing EGTA and centrifuged at 14,000×g for 10 min at 4°C. After resuspension in isolation buffer containing EGTA, the crude mitochondrial fraction was positioned on a discontinuous Ficoll gradient (F5415 Ficoll solution Type 400, 20% in H2O, Sigma, St. Louis, MO). The gradient consisted of two layers, with 2 mL of 7.5% on top of 2 mL of 10% Ficoll cut with isolation buffer containing EGTA, and centrifuged for 30 minutes at 100,000×g. The extrasynaptic mitochondrial pellet (bottom) and synaptic mitochondria trapped in synaptosomes at the interphase of the two Ficoll layers were the two distinct mitochondrial fractions created by the gradient. Collecting synaptosomes in 2.5 mL tubes, they were resuspended and centrifuged at 14,000×g for 10 min to remove Ficoll from the sample. Next, they were nitrogen decompressed in a cell disruption bomb manufactured by Parr Instrument Company (Moline, IL) to release mitochondria, and then they were cooled to 4°C under 1200 psi pressure for 10 min. In addition to being collected in 500 μL tubes, the extrasynaptic mitochondrial pellet was also resuspended and cleaned using isolation buffer containing EGTA. After being repositioned on a fresh Ficoll gradient, the damaged synaptosomes were put back into the ultracentrifuge and spun for 30 minutes at 4°C at 100,000×g. To remove the calcium chelator (EGTA), both the synaptic and extrasynaptic mitochondrial fractions were resuspended in isolation buffer without it. The mixture was then centrifuged at 14,000×g for ten minutes. To achieve a concentration of 10 mg/mL or more, the final mitochondrial pellets were resuspended in isolation buffer devoid of EGTA. Mitochondrial protein was measured using a microplate reader (Biotek, USA) and colorimetric methods [24].
Measurement of mitochondrial function
An Oxytherm Clark-type oxygen electrode (OXYT1/ED, Hansatech Instruments, Norfolk, UK) was used to measure mitochondrial function. The respiration buffer (125 mM KCl, 0.1% BSA, 20 mM HEPES, 2 mM MgCl2, 2.5 mM KH2PO4, pH 7.2) was added to the sealed Oxytherm chamber containing mitochondria (180-200 µg), which were then constantly agitated at 37ºC. Evaluations were conducted on various mitochondrial respiration states, including s-2, s-3, s-4, and s-5 complex-I and s-5 complex-II. The oxygen consumption from state 3 (ADP presence) to state 4 (oligomycin presence) was used to calculate the respiratory control ratio.
Mitochondrial calcium measurements
According to Korde et al., [23], mitochondrial Ca2+ cycling/ overloading was evaluated in situ [23]. Mitochondria were prepared according to the above instructions, except that the isolation buffer was Ca2+free and contained the following: (i) 0.6 μm ruthenium red to block the uniporter and the sodium-independent anti-porter of the mitochondria, (ii) 10 μm CGP-37157 to block the outward flux of Ca2+ via the sodium-dependent antiporter, and (iii) 5 μm Cyclosporin A to prevent Ca2+ loss through induction of the permeability transition. The isolation buffer does contain calcium chelators as well. Channel blockers were prepared in DMSO stock solutions with a minimum of 100×.50 μg of mitochondria were inserted into each well of a 96-well plate, and 5 μm Calcium Green 5-N was added, resulting in a final volume of 100 μL of “locking buffer.” The calcium levels were measured by measuring the fluorescence using a BIO-teck Synergy HT fluorometer with an excitation wavelength of 485 nm and an emission wavelength of 532 nm, both before and after solubilization with 100 μm Triton X-100 for 15 minutes. In order to extrapolate our fluorescence data to concentrations of [Ca2+]/mg mitochondrial protein, a standard Ca2+ curve with a range of 100 nM to 100 μM was created. The same total quantities were used for each test and standard. Mitochondrial protein was measured using a microplate reader (Biotek, USA) and colorimetric methods [24]. Fluorescent units are used to present values.
Mitochondrial permeability transition (MPT)
As previously mentioned, MPTP was determined as a result of measuring the swelling and contraction of the mitochondria using light scattering to assess the integrity of the mitochondrial membrane [46]. The protein concentration was used to standardize the change in absorbance at 520 nm.
Mitochondrial oxidative stress
Estimation of LPO and NO levels
The standard approach [42]. was followed in measuring the amount of mitochondrial Malondialdehyde (MDA). Micromoles of MDA/mg protein were used to express the degree of Lipid Peroxidation (LPO). Greiss reagent (0.1% at 540 nm) was used in a colorimetric experiment to measure the levels of Nitrite (NO); the results were expressed as nanomoles of NO/mg protein [14].
Estimation of mitochondrial SOD and CAT activity
The decrease of NBT at 560 nm in the presence of phenazine methosulphate and NADH was used to measure the activity of Superoxide Dismutase (SOD) [22]. Under the assay conditions, one unit of the enzyme was represented as 50% inhibition of NBT reduction per minute per mg protein. The measurement of catalase activity involved adding 50 milliliters of 6 percent H2O2, and then monitoring the absorbance drop at 240 nm for three minutes at 30-second intervals [1].
Analysis for cytoplasmic cytochrome-C, caspase-9, caspase-3
The Elisa test for caspase-3 (elabscience, USA, Cat# E-ELR0160) caspase-9 (elabscience, USA, Cat# E-EL-R0163) and cytochrome-c (elabscience, USA, Cat# E-EL-R0006) were performed as per manufacturer instruction. The brain tissue isolated from pups was weighed and minced into small pieces. The tissues were then homogenized in PBS (1:9 W/V) with a glass homogenizer on ice. The homogenate was centrifuged for 10 minutes at 5000g at 4℃, and the supernatant was collected. The 100 µl of standards and samples were incubated for 90 minutes at 37℃. After decanting the liquid, immediately add 100 µl of Biotinylated Detection Ab working solution to each well for 1 hour. The solution was aspirated and washed three times before being incubated with 100 µl HRP Conjugate working solution for 30 minutes. After that, 90µl of substrate solution was added in each well and incubated for 15 minutes. Finally, add 50 µl Stop Solution to each well. The optical density was determined using a microplate reader set to 450 nm.
Behavioral studies
Open field test (OFT)
The OFT was utilized to investigate variations in rearing behavior, time spent in the central grid, and spontaneous locomotor activity (ambulation). The experimental apparatus is an open-field square of 55×44×50 cm. The apparatus’s floor was separated into 12 fields and painted a dark grey color [35]. Every rat was positioned separately in the middle of the openfield device. The five minutes of the test were spent. After every test, ethanol was used to clean the equipment.
Elevated plus maze (EPM)
Animals were put on an EPM, as previously described, in order to gauge each person’s unique degree of anxiety-like behavior [44]. The device has two arms that are open (30×5×0.25 cm) and two arms that are closed (30×5×15 cm), both of which extend from a single central platform (5×5 cm). The entire equipment was raised in a room with low lighting (red light) to a height of 40 cm above the ground. Each mouse was given five minutes to freely explore the maze after being positioned in the center of it with its head facing a closed arm at the start of the session. Putting all four feet into one arm was the definition of an entrance. Anxiety is thought to be measured by the amount of time spent in open arms as opposed to closed arms [10,30]. Following every animal test, the plus-maze was meticulously cleaned with ethanol. Every training and testing session took place between 8:00 and 14:00 hours during the light phase.
Y-Maze test
A Y-maze paradigm was used to study animals’ spatial recognition memory [16]. The device was three black plastic arms, each measuring 50×10×20 cm3 and spaced 120 degrees apart, that were positioned in a room with visual clues painted on the walls. The Y-maze test included two trials with a one-hour gap between them. For five minutes during the first trial, the animal was kept at the end of one arm and had access to both arms. A door that resembled a guillotine stopped the third arm, or the new arm. After that, the rat was taken out of the labyrinth and put back in its own cage. The rat was returned to the starting arm of the maze for the second trial, and it had unrestricted access to all three arms for five minutes. Both the quantity of entries and the amount of time spent in every arm were noted. The proportion of entrances and time spent in the novel arm of the maze was contrasted with a haphazard tour of its three arms [2,31].
Forced swim test (FST)
The two sessions of the forced swimming test were a pretest (15 minutes) and the test (5 minutes), which was given 24 hours apart. Each animal was tested separately and kept in a transparent Plexiglas cylinder with a diameter of 18 cm and a height of 40 cm that was filled with 25°C water. Typically, during a pretest session, inexperienced rodents struggle to get away. But because the tank is unavoidable, an unsuspecting animal eventually develops a passive “despair” behavior (also known as “learned helplessness”) marked by a decrease in vigorous activity and an increase in immobile posture, which enables them to float by making only the necessary movements. This measurement is seen as an indicator of depression-like behavior because antidepressant medications shorten the immobility time [25,32]. The immobility time collected throughout the test session was analyzed to evaluate the animals’ depressive-like behavior.
Plasma corticosterone
The plasma CORT was quantified by a High-Performance Liquid Chromatography (HPLC) with Ultraviolet (UV) detector system (Waters, USA), according to Woodward and Emery (1987) and also as described by Prajapati and Krishnamurthy (2021), [2] (Woodward and Emery, 1987).
Statistical analysis
The means ± SEM are used to express the data. With regard to the analysis of cytochrome-C, caspase9, and caspase-3, mitochondrial bioenergetics, mitochondrial RCR, antioxidant enzyme, mitochondrial complex enzyme system, MMP, and MPT There was a two-way ANOVA and a Bonferroni post hoc test. Using a repeated measure two-way ANOVA and a Bonferroni post hoc test, sensorimotor behavioral activities were measured. P<0.05 was regarded as significant. GraphPad Prism version 8.0.2 for Windows (GraphPad Software, San Diego, CA, USA) was used to analyze all of the data.
Results
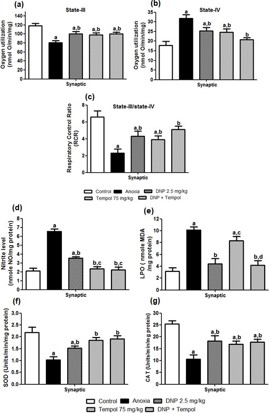
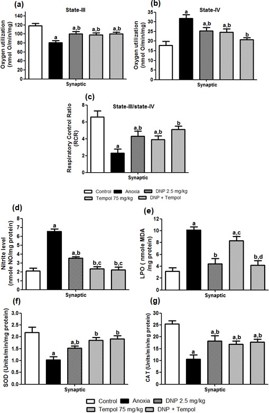
Effect of 2,4 DNP, tempol and their combination (D+ T) on anoxia-induced changes in synaptic mitochondrial s-III and s-IV respiration and RCR in cortical brain region on d-7
Figure 1(a-c) shows the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in synaptic mitochondrial s-III (Figure 1(a)) respiration and s-IV (Figure 1(b)) respiration and RCR (Figure 1(c)) in cortical brain region on d-7. An ANOVA depicted a significant main effect for s-III [F (4,20) = 7.129; P<0.05], s-IV [F (4,20) = 9.232; P<0.05] and respiration and RCR [F (4,20) = 8.607; P<0.05]. Post hoc analysis revealed that treatment with 2,4 DNP, tempol and their combination (D+T) was significantly effective in treating s-III, s-IV respiration and RCR compared to anoxia group animals. Further, there was no significant difference between the combination (D+T) or individual drug treatment (2,4 DNP or tempol respectively).
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in synaptic mitochondrial NO, LPO, SOD and CAT levels in cortical brain region on d-7
Figure 1(d-g) illustrates the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in synaptic mitochondrial Nitric oxide (Figure 1(d)), LPO (Figure 1(e)), SOD (Figure 1(f)) and CAT (Figure 1(g)) in the cortical brain region. One way ANOVA revealed a significant difference in the levels of NO [F (4,20) = 47.92; P<0.05], LPO [F (4,20) = 18.12; P<0.05], SOD [F (4,20) = 8.78; P<0.05] and CAT [F (4,20) = 10.85; P<0.05] post-anoxia injury. Post hoc analysis showed that treatment with 2,4 DNP, tempol and their combination (D+T) was significantly effective in attenuating NO, SOD and CAT levels compared to anoxia group animals. However, 2,4 DNP, and the combination (D+T) but not tempol reduce the LPO levels. Further, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol).
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in synaptic mitochondrial Ca2+ and MPT in cortical brain region on d-7
Figure 2(a,b) represents the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in synaptic mitochondrial (Figure 2(a)) calcium overload (Figure 2(b)) MPT in cortical brain region on day-7. An ANOVA revealed a significant difference in calcium overload [F (4,20) = 16.98; P<0.05] and synaptic –mitochondrial MPT in terms of mitochondrial swelling [F (4,20) = 7.34; P<0.05] post-anoxia injury. Post hoc analysis showed that treatment with 2,4 DNP and the combination (D+T) was significantly effective in decreasing synapticmitochondrial calcium overloads. However, tempol was not found effective in decreasing the same. Further, 2,4 DNP, tempol and the combination (D+T) was found effective in mitigating mitochondrial swelling compared to anoxia group animals. Furthermore, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol).
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in cytochrome-C, caspase-9 and caspase-3 in synaptic cortical brain region on d-7
Figure 2 shows the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in cytochrome-C (Figure 2(c)), caspase-9 (Figure 2(d)) and caspase-3 (Figure 2(e)) on day-7. One way ANOVA revealed a significant main effect for cytochrome-C [F (4,10) = 29.6, P<0.05], caspase-9 [F (4,10) = 18.2, P<0.05] and caspase-3 [F (4,10) = 18.2, P<0.05] post-anoxia. Post hoc analysis showed that treatment with 2,4 DNP, tempol and the combination (D+T) was significantly effective in decreasing the levels of cytochrome-C, caspase-9 and caspase-3 compared to anoxia group animals. However, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol).
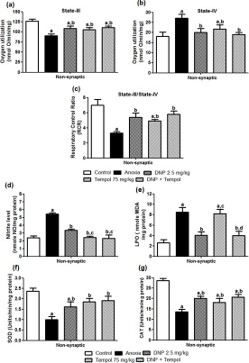
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in non-synaptic mitochondrial s-III and s-IV respiration and RCR in cortical brain region on d-7
Figure 3(a-c) shows the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in non-synaptic mitochondrial s-III respiration (a) and s-IV respiration (b) and RCR (c) in cortical brain region on d-7. An ANOVA depicted a significant main effect for s-III [F (4,20) = 6.67; P<0.05] and s-IV [F (4,20) = 3.48; P<0.05] respiration and RCR [F (4,20) = 8.49; P<0.05]. Post hoc analysis revealed that treatment with 2,4 DNP, tempol and their combination (D+T) was significantly effective in treating s-III, s-IV respiration, and RCR compared to anoxia group animals. Further, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol).
Effect of 2,4 DNP, tempol and their combination (D+ T) on anoxia-induced changes in non-synaptic mitochondrial Nitric oxide, LPO, SOD, and CAT in cortical brain region on d-7
Figure 3(d-g) depicts the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in non-synaptic mitochondrial Nitric oxide (d), LPO (e), SOD (f) and CAT (g) in the cortical brain region. One-way ANOVA revealed a significant decrease in the levels of NO [F (4,20) = 28.10; P<0.05], LPO [F (4,20) = 11.20; P<0.05], SOD [F (4,20) = 7.32; P<0.05] and CAT [F (4,20) = 16.75; P<0.05] following anoxia. Post hoc analysis showed that treatment with 2,4 DNP, tempol and their combination (D+T) was significantly effective in attenuating NO, SOD and CAT levels compared to anoxia group animals. However, 2,4 DNP, and the combination (D+T) but not tempol reduce the LPO levels. Further, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol). Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in non-synaptic mitochondrial calcium and MPT in cortical brain region on d-7
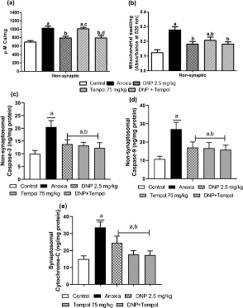
Figure 4(a,b) represent the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in nonsynaptic mitochondrial calcium overload (Figure 4(a)) and MPT (Figure 4(b)) in cortical brain region on day-7. An ANOVA revealed a significant anoxia-induced increase in non-synaptic mitochondrial calcium overload [F (4,20) = 10.98; P<0.05] and increase in non-synaptic mitochondrial swelling [F (4,20) = 7.79; P<0.05] post-anoxia injury. Post hoc analysis showed that treatment with 2,4 DNP and the combination (D+T) was significantly effective in decreasing non-synaptic-mitochondrial calcium overloads. However, tempol was not found effective in decreasing the same. Further, 2,4 DNP, tempol and the combination (D+T) was found effective in mitigating mitochondrial swelling compared to anoxia group animals. Furthermore, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol).
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in cytochrome-C, caspase-9 and caspase-3 in non-synaptic cortical brain region on d-7
Figure 4(c-e) shows the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in non-synaptic cytochrome-C (Figure 4(c)), caspase-9 (Figure 4(d)) and caspase-3 (Figure 4(e)) on day-7. One way ANOVA revealed a significant main effect for cytochrome-C [F (4,10) = 29.6, P<0.05], caspase-9 [F (4,10) = 18.2, P<0.05] and caspase-3 [F (4,10) = 18.2, P<0.05] post-anoxia. Post hoc analysis showed that treatment with 2,4 DNP, tempol and the combination (D+T) was significantly effective in decreasing the levels of cytochrome-C, caspase-9 and caspase-3 compared to anoxia group animals. Furthermore, there was no significant difference between the combination (D+T) and individual drug treatment (2,4 DNP or tempol).
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced alterations in spontaneous locomotor activity in OFT
The effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in ambulation, time spent in the central grid and rearing in OFT are depicted in Table 1. A repeated measure two-way ANOVA depicted a significant difference in ambulation among groups [F (4,100) = 106.1, P<0.05], time [F (4,100) = 11.8, P<0.05]. There was no significant interaction between group and time among groups [F (16,100) = 0.38, P>0.05]. A post hoc analysis showed that there was a marked increase in ambulatory behavior in anoxia group from d-21 to d-150 compared to control group animals. However, treatment with 2,4 DNP, tempol or their combination (D+T) was significantly (p<0.05) effective in decreasing number of ambulations on different days. Similarly, an ANOVA depicted a significant main effect for time spent in central grid and rearing among groups ([F (4,100) = 9.26, P<0.05 and F(4,100) = 20.13, P<0.05] respectively), time ([F (4,100) = 22.0, P<0.05 and F(4,100) = 10.15, P<0.05] respectively) and a significant interaction between group and time ([F (16,100) = 4.14, P<0.05 and F(16,100) = 3.72, P<0.05] respectively). A post hoc analysis showed that there was no significant difference (p>0.05) for time spent in the central grid and rearing in all days between control and anoxia groups.
Table 1: 2,4 DNP, tempol and their combination (D+T) on anoxia-induced changes in ambulation, time spent in central grid and rearing during OFT.
| Day | Control | Anoxia | DNP | Tempol | DNP+Tempol |
|---|---|---|---|---|---|
| Ambulation (in number) | |||||
| 21 | 63.7±7.41 | 96.5±5.83* | 85.6±4.51*, # | 82.6±6.81*, # | 83.6±4.62*, # |
| 60 | 65.6±8.24 | 98.2±6.23* | 85.4±6.61*, # | 88.4±7.29*, # | 80.4±5.21*, # |
| 90 | 61.9±8.71 | 97.1±5.37* | 84.5±3.26*, # | 85.3±6.41*, # | 76.5±6.40# |
| 120 | 55.9±4.45 | 88.3±6.61* | 76.3±7.23*, # | 75.5±4.32*, # | 70.3±5.33*, # |
| 150 | 60.5±5.24 | 94.5±8.38* | 82.4±3.62*, # | 84.3±7.21*, # | 79.4±4.56*, # |
| Time spent in central grid (in sec) | |||||
| 21 | 13.5±4.21 | 11.5±2.24 | 12.6±1.93 | 9.4±1.87* | 9.2±2.33*, ** |
| 60 | 12.6±1.40 | 15.4±1.62 | 17.3±1.13* | 19.6±1.40* | 12.9±1.13**, ## |
| 90 | 11.8±1.23 | 13.2±1.13 | 13.5±2.22 | 12.9±1.39# | 10.5±3.11 |
| 120 | 13.5±1.45 | 16.5±1.85 | 11.1±2.36# | 13.7±1.15 | 10.3±2.24#, ## |
| 150 | 11.6±1.12 | 12.1±2.56 | 10.5±1.45 | 9.7±1.23 | 9.6±3.13 |
| Rearing (in number) | |||||
| 21 | 11.5±3.67 | 15.2±2.25 | 18.4±1.88* | 20.1±3.33*, # | 26.7±2.12*, #, **, ## |
| 60 | 12.6±1.32 | 14.2±1.49 | 16.1±2.63 | 15.6±1.47 | 13.5±3.14 |
| 90 | 14.8±4.11 | 17.8±4.19 | 19.5±2.29* | 19.9±2.34* | 20.5±3.34* |
| 120 | 15.1±2.47 | 16.1±1.90 | 18.8±4.61 | 20.1±3.25* | 18.1±2.26 |
| 150 | 14.2±1.14 | 12.1±2.44 | 18.2±2.65# | 19.7±1.29*, # | 15.8±3.19 |
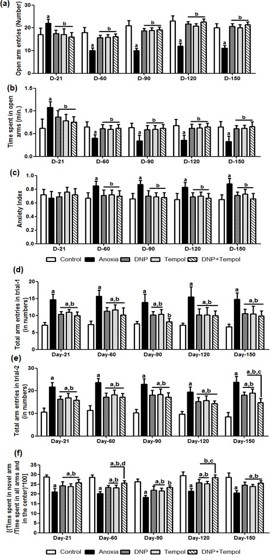
Effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced anxiety-like behavior in EPM test
Figure 5(a-c) depicts the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced alterations in open arm entries (a) time spent in open arms (b) and anxiety index (c) in elevated plus-maze test paradigm. A repeated measure twoway ANOVA depicted a significant differences in open arm entries and time spent in open arms among groups ([F (4,100) = 71.93, P<0.05 and F (4,100) = 9.96, P<0.05] respectively), time ([F (4,100) = 30.38, P<0.05 and F (4,100) = 26.13, P<0.05] respectively) and a significant interaction between group and time ([F (16,100) = 16.58, P<0.05 and F(16,100) = 6.98, P<0.05] respectively). A post hoc analysis showed that there was a marked decrease in open arm entries and time spent in open arms from d-21 up to d-150. Treatment with 2, 4 DNP, tempol or their combination (D+T) was effective in increasing open arm entries and open arm residing time. Similarly, a significant main effect for anxiety index among groups [F (4,100) = 16.02, P<0.05], insignificant with time [F (4,100) = 0.388, P>0.05], While, a significant interaction between group and time [F (16,100) = 2.1, P<0.05] was observed. However, there was no effect of anoxia on anxiety index on d-21. A marked increase in anxiety index was observed from d-60 to d-150. 2, 4 DNP, tempol or their combination (D+T) attenuated anxiety-like behavior in EPM task from d-60 to d-150 post-anoxia injury. It is interesting to note that there is no significant difference in D+T as compared to either 2,4 DNP or tempol.
Effect of 2,4 DNP, tempol and their combination (D+T) attenuated anoxia-induced behavioral alterations in Y-maze test
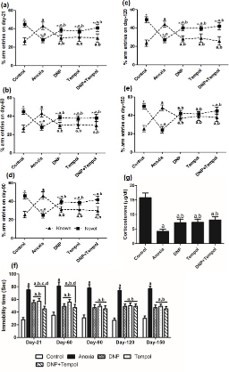
The effect of anoxia-induced alterations in general exploratory behavior (curiosity) in trial-1 and trial-2 are depicted in Figure 5(d,e) respectively. Figure 5(f) shows the coping behavior to novel arm (anxiety-like behavior). A repeated measure two-way ANOVA depicted a significant difference for exploration in trial-1 and trial-2 among groups ([F (4,100) = 80.43; P<0.05] and [F (4,100) = 133.6; P<0.05] respectively), time ([F (4,100) = 1.87; P<0.05] and [F (4,100) = 7.90; P<0.05] respectively). However, no significant interaction between group and time ([F (16,100) = 0.47; P>0.05] and [F (16,100) = 1.27; P>0.05] respectively) was observed. Similarly, there were significant changes in coping behavior among groups [F (4,100) =1 04.4; P<0.05], time [F (4,100) = 22.40; P<0.05]. However, there was no significant interaction between group and time [F (16,100) = 0.44; P>0.05]. Post hoc analysis revealed that anoxia leads to a marked increase in exploration regarding hyperactivity both trial-1 and trial-2 and copying behavior from d-21 up to d-150. However, 2,4 DNP, tempol and their combination (D+T) significantly reversed anoxia-induced increase in hyper activeness both in trial-1 and trial-2 and decreased in coping behavior from d-21 up to d-150. However, there was no synergistic effect observed in the combination (D+T) as compared to individual effects of drugs.
Further, in Figure6(a-e), the effect of 2,4 DNP, tempol and their combination (D+T) on anoxia-induced alterations in spatial recognition memory on d-21 (a), d-60 (b), d-90 (c), d-120 (d), d-150 (e) are depicted. A repeated measure two-way ANOVA showed significant differences in arm discrimination behavior on d-21, d-60, d-90, d-120 and d-150 during trial-2 among groups [F (4,40) = 0.41; P<0.05, F (4,40) = 0.55; P<0.05, F (4,40) = 1.64; P<0.05], [F (4,40) = 0.87; P<0.05] and [F (4,40) = 0.33; P<0.05 respectively], significant main effect for known and novel arms ([F (1,40) = 32.10; P<0.05], [F (1,40) = 25.95; P<0.05], [F (1,40) = 28.50; P<0.05], [F (1,40) = 86.32; P<0.05] and [F (1,40) = 9.08; P<0.05 respectively) and a significant interaction between group and arms ([F (4,40) = 28.35; P<0.05], [F (4,40) = 27.26; P<0.05], [F (4,40) = 45.25; P<0.05], [F (5,40) = 48.34; P<0.05] and [F (4,40) = 58.09; P<0.05] respectively in Y-maze test paradigm. Post-hoc analysis revealed that there was a marked priority to novel arm entries for control group animals on different days. A Within group comparison further depicted that anoxia caused significant decrease in novel arm preference compared to known arm on d-21 to d-150. 2,4 DNP, tempol and their combination (D+T) attenuated the anoxia-induced decrease in novel arm entries from d-21 itself which was further maintained up to d-150. Furthermore, repeated measure two-way ANOVA revealed significant differences for known and novel arm entries among groups ([F (4,100) = 81.59; P<0.05] and [F (4,100) = 128.3; P<0.05] respectively) time ([F (4,100) = 11.89; P<0.05] and [F (4,100) = 3.67; P<0.05] respectively) and a significant interaction between group and time ([F (4,100) = 1.12; P<0.05] and [F (4,100) = 1.38; P<0.05] respectively). Post-hoc analysis revealed that anoxia increased the percentage entries into known arm compared to control group animals from d-21 to d-150. 2,4 DNP, tempol and their combination (D+T) increased the percentage entries into novel arm in all days compared to anoxia group. However, there was no synergistic effect observed in the combination (D+T) compared to individual effect of 2,4DNP and tempol. Likewise, while considering novel arm entries, anoxia significantly decreased the percentage entries into novel arm compared to control group animals from d-21 up to d-150. 2,4 DNP, tempol and their combination (D+T) markedly reversed the anoxia-induced decrease in the percentage entries into novel arm on different days. Although, there was no significant difference among 2,4 DNP, tempol and their combination (D+T) in both known and novel arm entries on all days.
DNP, tempol and their combination (D+T) improved anoxia-induced depression-like symptoms in FST
Figure 6f shows the effect of 2,4 DNP (2.5 mg/kg), tempol (75 mg/kg) and their combination (D+T) on anoxia-induced immobility period in FST on different days of experimental design. Repeated measure two-way ANOVA revealed a significant main effect among groups [F (4,100) = 374.3; P<0.05], time [F (4,100)= 4.0; P<0.05] and a significant interaction between group and time [F (16,100) = 2.09; P<0.05]. Post hoc analysis showed that anoxia caused a markedly increased the immobility period compared to control animals in all days. Treatment with 2,4 DNP, tempol and their combination (D+T) altered anoxia-induced increase in immobility period on d-21up to d-150. The effectiveness of combination (D+T) was similar to the individual effect of 2,4 DNP and tempol.
Effect of 2,4 DNP, tempol and their combination (D+T) on plasma corticosterone level
The effect of 2,4 DNP (2.5 mg/kg), tempol (75 mg/kg) and their combination (D+T) on anoxia-induced alterations in plasma CORT level is shown in Figure 6g. Statistical analysis by oneway ANOVA depicted a significant difference in plasma CORT levels among groups [F (4,20) = 66.46; P<0.05] on d-150. Posthoc analysis revealed that anoxia caused a significant decrease in the level of CORT in plasma compared to control animals. 2,4 DNP, tempol and D+T significantly increased anoxia-induced decrease in plasma CORT level. The combination of both drugs (D+T) was similarly effective as to their individual effects.
Discussion
This is the first study showing the combination effect of mitochondrial modulators like 2,4 DNP and tempol to reduce neuronal insult progression by improving synaptic and non-synaptic mitochondrial function in developing brain of and further a long-term improvement in neurobehavioral changes in later days of life using a global model of anoxia. Overall, there was no synergistic effect observed while treatment with the combination of 2,4 DNP and tempol.
Anoxia hampered the mitochondrial respiration in both synaptic and non-synaptic mitochondrial fractions in respect to altered mitochondrial oxygen consumption in s-3 and s-4. Further, a marked decrease in RCR was also observed in the two different mitochondrial fractions which depict compromised mitochondrial bioenergetics. Treatment with the combination of D+T was equally effective to their individual effects in improving s-3, s-4 as well as RCR in both mitochondrial fractions. Fur-ther, the combination of D+T was equally effective as compared to the individual effect of 2,4 DNP and tempol in decreasing anoxia-induced levels of NO and LPO in both the mitochondrial fractions. Our previous finding also sows the decrease in NO and LPO levels with respect to 2,4 DNP [40] and tempol [39]. Further, the levels of antioxidant defense system like SOD and CAT activity were hindered by anoxic injury. The combination of D+T significantly improved the antioxidant defense system in both synaptic and non-synaptic mitochondrial fractions similar to the individual effects of both drugs. In normal physiological conditions, mitochondria act as a sink to maintain Ca2+ homeostasis. During pathological conditions like anoxia, mitochondria become overloaded with Ca2+ and undergo the cataclysmic Mitochondrial Permeability Transition (MPT) formation within the inner mitochondrial membrane with a resultant rupture of the outer mitochondrial membrane caused by osmotic swelling [3]. We observed a marked increase in the levels of Ca2+ concentrations in both synaptic and non-synaptic mitochondria. However, there was no synergistic effect observed on treatment with D+T as compared to 2,4 DNP in mitigating Ca2+ overloads in both the mitochondrial fractions. This effect of 2,4 DNP may be due to its mild uncoupling effect. However, tempol failed to elicit any protective effect in reducing the Ca2+ loads. Similarly, anoxia caused a significant increase in both synaptic and non-synaptic mitochondrial swelling. The combination of 2,4 DNP, tempol and their combination was equally effective in reducing the swelling in both fractions. The translocation of Bax to the mitochondrial outer membrane from cytosol induces Mitochondrial Outer Membrane Permeabilization (MOMP) which along with inner membrane lead to the formation of mPTP and causes the leakage of apoptogenic factors such as cytochrome-C through the mitochondrial intermembrane space proteins [9,15,21]. Furthermore, anoxia leads to a marked increase in the expression of cytochrome-c, caspase-9 and caspase-3. The combination of D+T significantly mitigated the expression of these apoptotic proteins and cytochrome-C.
Anoxic injury in newborns causes delayed behavioral disturbances such as abnormal responses to stress and impaired learning which persists in a lifelong manner [6]. The results of the present investigation confirm the existence of abnormal responses to stress in adolescent rats previously subjected to neonatal anoxia. OFT is used to evaluate the spontaneous locomotor activity which can be considered as a sign of hyperactivity in animals. Anoxia caused a significant increase in the spontaneous locomotor activity regarding ambulation from d-21 to d-150. A previous report has suggested that the augmented locomotor activity is likely to be due to disturbances in the development of brain regions such as the hippocampus, corpus striatum or amygdala occurring during a specific postnatal time window [33]. However, no marked changes in the time spent in central grid and rearing was observed from d-21 to d-150. 2,4 DNP, tempol and their combination (D+T) mitigated the ambulation behavior depicting the attenuation of hyperactiveness in rats from d-21 to d-150. Both experimental and clinical studies suggest that adverse experience during development augments depression and anxiety in adulthood [11,18,45]. It has been suggested that the decrease in open arm entries and time spent in open arm and anxiety index are the indicators of anxiety-like activity in EPM [28,44]. In our study, anoxia caused a marked increase on d-21 and further decrease from d-60 to d-150 in the number of open arm entries and time spent in the open arm on d-21. However, there were no significant changes in the anxiety index on d-21. Further, a marked increase in anxiety index was observed from d-60 to d-150. The increase in open arm entries and open arm time can be predicted as a sign of hyperactivity of rats. The combination of D+T was equally effective in decreasing the open arm entries and open arm time on d-21 and further increasing the same along with decreasing the anxiety index from d-60 to d-150 similarly as 2,4 DNP and tempol. This shows that 2,4 DNP, tempol and D+T decreased the anxiety-like behavior as an effect of anoxia and improved the neurobehavioral outcome.
Anoxia caused marked alterations in the trial-1 and trial-2 regarding exploration (curiosity), time spent in the novel to all arms as a measure of anxiety index (coping) and percentage is known to novel arm entries as a measure of spatial recognition memory in Ymaze paradigm from d-21 to d-150. 2,4 DNP, tempol and D+T combination markedly attenuated the hyperactive by reducing curiosity in trial-1 and trial-2, reduction in coping behavior and increase in the novel to known arm entries as a measure of improved spatial recognition memory from d-21 to d-150. A previous report has shown that neonatal anoxia leads to disruption of reference spatial memory associated with impairments in the spatial working memory in adulthood [45]. The spatial deficits post neonatal anoxia may be the result of damage to neuronal systems directly involved in cognitive or attentional processes [5]. Another aspect of emotionality that is affected by neonatal anoxia is depression-like behavior, which is typically identified either by expression of gustatory anhedonia or behavioral despair [25]. There was a marked increase in the immobility time in FST from d-21 up to day-150 as compared to control group animals. We observed a marked attenuation of immobility time on treatment with 2,4 DNP, tempol and D+T from d-21 to d-150.
Postnatal stress in rodents caused by neonatal anoxia increases emotionality, an effect that tends to be associated with HPA axis dysregulation [17]. The stress-induced corticosterone hyposecretion was also observed in adult animals born by caesarean section applied after experimental anoxia [33]. We observed marked decrease in the levels of plasma corticosterone. 2,4 DNP, tempol and D+T markedly corrected the corticosterone levels and attenuated the HPA axis activity on day-150.
We conclude that the combination of 2,4 DNP and tempol showed an additive effect on individual drugs in improving mitochondrial function as well as neurobehavioral outcomes postanoxia injury.
Author declarations
Funding: SK and PKS is thankful to Department of Biotechnology (DBT), New Delhi, India for assistance regarding research grant [102/IFD/SAN/4654/2011-2012].
Author’s contributions and statement: SK and PKS designed the study and wrote the manuscript. PKS and SKP performed the experiment and analysed the results. AK assisted in data analysis. We declared that this manuscript is original, has not been published before and is not currently being considered for publication elsewhere.
Data availability: Data will be made available on request.
Author statement: All animal experiments were performed in accordance with the National Institute of Health Guidelines (publication number 85-23, revised 2013). Experiments on animals were approved by the Institutional Animal Ethical Committee, BHU, Varanasi, India (Protocol No. Dean/11-12/CAEC/328).
Declaration of competing interests: The authors report no potential conflicts of interest.
References
- Beers RF Jr, Sizer IW. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem. 1952; 195: 133-40.
- Bhattacharjee A, Prajapati SK, Krishnamurthy S. Supplementation of taurine improves ionic homeostasis and mitochondrial function in the rats exhibiting post-traumatic stress disorder-like symptoms. Eur J Pharmacol. 2021; 908: 174361.
- Brown MR, Sullivan PG, Geddes JW. Synaptic mitochondria are more susceptible to Ca2+ overload than nonsynaptic mitochondria. J Biol Chem. 2006; 281: 11658-68.
- Burke RE, Macaya A, DeVivo D, Kenyon N, Janec EM. Neonatal hypoxic-ischemic or excitotoxic striatal injury results in a decreased adult number of substantia nigra neurons. Neuroscience. 1992; 50: 559-69.
- Buwalda B, Nyakas C, Vosselman HJ, Luiten PGM. Effects of early postnatal anoxia on adult learning and emotion in rats. Behav Brain Res. 1995; 67: 85-90.
- Caputa M, Rogalska J, Wentowska K, Nowakowska A. Perinatal asphyxia, hyperthermia and hyperferremia as factors inducing behavioural disturbances in adulthood: A rat model. Behav Brain Res. 2005; 163: 246-56.
- Cerio FGd, Lara-Celador I, Alvarez A, Hilario E. Neuroprotective therapies after perinatal hypoxic-ischemic brain injury. Brain Sci. 2013; 3: 191-214.
- Charles MS, Ostrowski RP, Manaenko A, Duris K, Zhang JH, Tang J. Role of the pituitary-adrenal axis in granulocyte-colony stimulating factor-induced neuroprotection against hypoxia-ischemia in neonatal rats. Neurobiol Dis. 2012; 47: 29-37.
- Chipuk JE, Green DR. How do BCL-2 proteins induce mitochondrial outer membrane permeabilization? Trends Cell Biol. 2008; 18: 157-64.
- Cohen H, Matar MA, Buskila D, Kaplan Z, Zohar J. Early poststressor intervention with high-dose corticosterone attenuates posttraumatic stress response in an animal model of posttraumatic stress disorder. Biol Psychiatry. 2008; 64: 708-17.
- Fan JM, Chen XQ, Du JZ. Prenatal stress, anxiety and depression: a mechanism involving CRH peptide family. Neuro Endocrinol Lett. 2014; 35: 429-39.
- Fan LW, Lin S, Pang Y, Lei M, Zhang F, Rhodes PG, Cai Z. Hypoxiaischemia induced neurological dysfunction and brain injury in the neonatal rat. Behav Brain Res. 2005; 165: 80-90.
- Gilmer LK, Ansari MA, Roberts KN, Scheff SW. Age-related changes in mitochondrial respiration and oxidative damage in the cerebral cortex of the Fischer 344 rat. Mech Ageing Dev. 2010; 131: 133-43.
- Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem. 1982; 126: 131-38.
- Hagberg H, Mallard C, Rousset CI, Xiaoyang W. Apoptotic mechanisms in the immature brain: involvement of mitochondria. J Child Neurol. 2009; 24: 1141-46.
- Hansen F, Pandolfo P, Galland F, Torres FV, Dutra MF, Batassini C, et al. Methylglyoxal can mediate behavioral and neurochemical alterations in rat brain. Physiol Behav. 2016; 164: 93-101.
- Heim C, Nemeroff CB. The role of childhood trauma in the neurobiology of mood and anxiety disorders: preclinical and clinical studies. Biol Psychiatry. 2001; 49: 1023-39.
- Heim C, Shugart M, Craighead WE, Nemeroff CB. Neurobiological and psychiatric consequences of child abuse and neglect. Dev Psychobiol. 2010; 52: 671-90.
- Hill RL, Kulbe JR, Singh IN, Wang JA, Hall ED. Synaptic mitochondria are more susceptible to traumatic brain injury-induced oxidative damage and respiratory dysfunction than non-synaptic mitochondria. Neuroscience. 2018; 386: 265-83.
- Hill RL, Singh IN, Wang JA, Kulbe JR, Hall ED. Protective effects of phenelzine administration on synaptic and non-synaptic cortical mitochondrial function and lipid peroxidation-mediated oxidative damage following TBI in young adult male rats. Exp Neurol. 2020; 330: 113322.
- Iriyama T, Kamei Y, Kozuma S, Taketani Y. Bax-inhibiting peptide protects glutamate-induced cerebellar granule cell death by blocking Bax translocation. Neurosci Lett. 2009; 451: 11-15.
- Kakkar P, Das B, Viswanathan PN. A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys. 1984; 21: 130-32.
- Korde AS, Pettigrew LC, Craddock SD, Maragos WF. The mitochondrial uncoupler 2,4-dinitrophenol attenuates tissue damage and improves mitochondrial homeostasis following transient focal cerebral ischemia. J Neurochem. 2005; 94: 1676-84.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951; 193: 265-75.
- Markostamou I, Ioannidis A, Dandi E, Mandyla MA, Nousiopoulou E, Simeonidou C, et al. Maternal separation prior to neonatal hypoxia-ischemia: impact on emotional aspects of behavior and markers of synaptic plasticity in hippocampus. Int J Dev Neurosci. 2016; 52: 1-12.
- Naga KK, Sullivan PG, Geddes JW. High cyclophilin D content of synaptic mitochondria results in increased vulnerability to permeability transition. J Neurosci. 2007; 27: 7469-75.
- Pandya JD, Pauly JR, Nukala VN, Sebastian AH, Day KM, Korde AS, et al. Post-injury administration of mitochondrial uncouplers increases tissue sparing and improves behavioral outcome following traumatic brain injury in rodents. J Neurotrauma. 2007; 24: 798-811.
- Patel B, Das SK, Das S, Das L, Patri M. Neonatal exposure to benzo[a]pyrene induces oxidative stress causing altered hippocampal cytomorphometry and behavior during early adolescence period of male Wistar rats. Int J Dev Neurosci. 2016; 50: 7-15.
- Petersen MH, Willert CW, Andersen JV, Waagepetersen HS, Skotte NH, Nørremølle A. Functional differences between synaptic mitochondria from the striatum and the cerebral cortex. Neuroscience. 2019; 406: 432-43.
- Prajapati SK, Ahmed S, Rai V, Gupta SC, Krishnamurthy S. Suvorexant improves mitochondrial dynamics with the regulation of orexinergic and mTOR activation in rats exhibiting PTSD-like symptoms. J Affect Disord. 2024; 350: 24-38.
- Prajapati SK, Chippala R, Krishnamurthy S. Inhibition of phosphodiesterase-4 mitigates stress-re-stress-paradigm induced mitochondrial perturbations in rats exhibiting PTSD-like symptoms. Neurosci Behav Physiol. 2023; 53: 1330-46.
- Prajapati SK, Dangi DS, Krishnamurthy S. Repeated caffeine administration aggravates post-traumatic stress disorder-like symptoms in rats. Physiol Behav. 2019; 211: 112666.
- Rogalska J, Caputa M. Neonatal asphyxia under hyperthermic conditions alters HPA axis function in juvenile rats. Neurosci Lett. 2010; 472: 68-72.
- Rogalska J, Kang P, Wotherspoon W, Macleod MR, Lai M. Effect of hyperthermia and anoxia on glucocorticoid and mineralocorticoid receptor expression in neonatal rat hippocampus. Neurosci Lett. 2009; 450: 196-200.
- Rojas JJ, Deniz BF, Miguel PM, Diaz R, Hermel EE, Achaval M, et al. Effects of daily environmental enrichment on behavior and dendritic spine density in hippocampus following neonatal hypoxia-ischemia in the rat. Exp Neurol. 2013; 241: 25-33.
- Samaiya PK, Krishnamurthy S. Characterization of mitochondrial bioenergetics in neonatal anoxic model of rats. J Bioenerg Biomembr. 2015; 47: 217-22.
- Samaiya PK, Krishnamurthy S, Kumar A. Mitochondrial dysfunction in perinatal asphyxia: role in pathogenesis and potential therapeutic interventions. Mol Cell Biochem. 2021; 476: 4421-34.
- Samaiya PK, Narayan G, Kumar A, Krishnamurthy S. Neonatal anoxia leads to time dependent progression of mitochondrial linked apoptosis in rat cortex and associated long term sensorimotor deficits. Int J Dev Neurosci. 2016; 52: 55-65.
- Samaiya PK, Narayan G, Kumar A, Krishnamurthy S. Tempol (4 hydroxy-tempo) inhibits anoxia-induced progression of mitochondrial dysfunction and associated neurobehavioral impairment in neonatal rats. J Neurol Sci. 2017; 375: 58-67.
- Samaiya PK, Narayan G, Kumar A, Krishnamurthy S. 2,4-Dinitrophenol attenuates mitochondrial dysfunction and improves neurobehavioral outcomes postanoxia in neonatal rats. Neurotox Res. 2018; 34: 121-36.
- Strackx E, Van den Hove DL, Prickaerts J, Zimmermann L, Steinbusch HW, Blanco CE, et al. Fetal asphyctic preconditioning protects against perinatal asphyxia-induced behavioral consequences in adulthood. Behav Brain Res. 2010; 208: 343-51.
- Sunderman F, Marzouk A, Hopfer S, Zaharia O, Reid M. Increased lipid peroxidation in tissues of nickel chloride-treated rats. Ann Clin Lab Sci. 1985; 15: 229-36.
- Sweis BM, Bachour SP, Brekke JA, Gewirtz JC, Sadeghi-Bazargani H, Hevesi M, et al. A modified beam-walking apparatus for assessment of anxiety in a rodent model of blast traumatic brain injury. Behav Brain Res. 2016; 296: 149-56.
- Sweis BM, Veverka KK, Dhillon ES, Urban JH, Lucas LR. Individual differences in the effects of chronic stress on memory: behavioral and neurochemical correlates of resiliency. Neuroscience. 2013; 246: 142-59.
- Takada SH, Motta-Teixeira LC, Machado-Nils AV, Lee VY, Sampaio CA, Polli RS, et al. Impact of neonatal anoxia on adult rat hippocampal volume, neurogenesis and behavior. Behav Brain Res. 2016; 296: 331-38.
- Tedeschi H, Harris DL. Some observations on the photometric estimation of mitochondrial volume. Biochim Biophys Acta. 1958; 28: 392-402.
- Weitzdoerfer R, Gerstl N, Pollak D, Hoeger H, Dreher W, Lubec G. Long-term influence of perinatal asphyxia on the social behavior in aging rats. Gerontology. 2004; 50: 200-5.